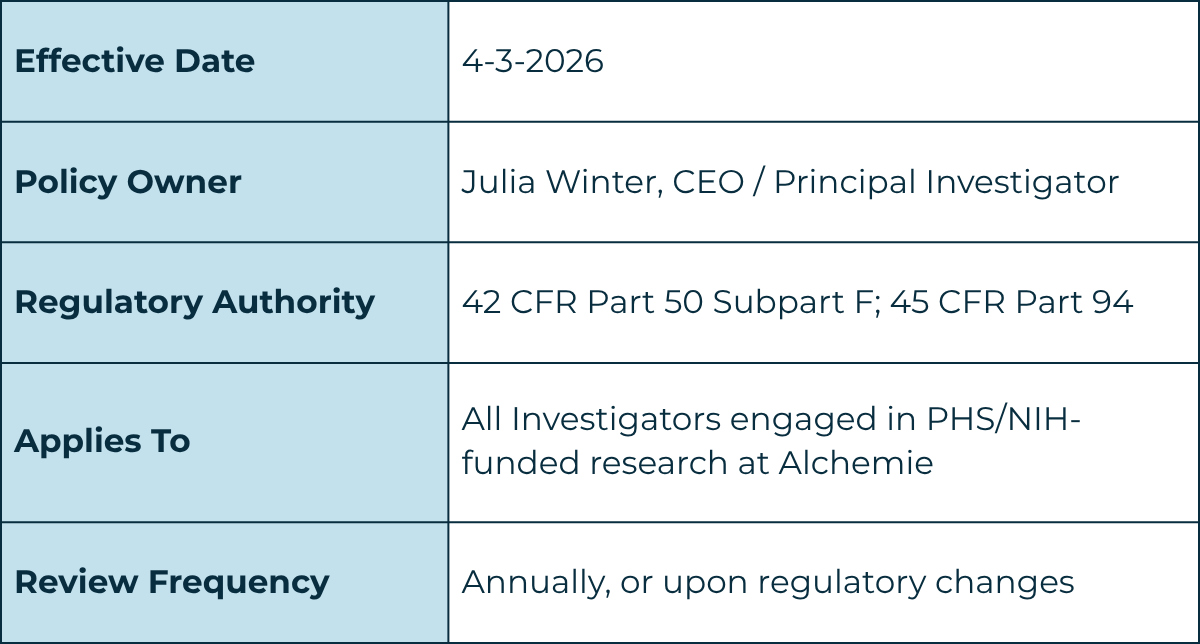
1. Introduction and Purpose
The purpose of this policy is to ensure that research funded by the National Institutes of Health (NIH) / Public Health Service (PHS) is designed, conducted, and reported objectively and without bias resulting from Investigator financial conflicts of interest (FCOI).
This policy implements the requirements of 42 CFR Part 50 Subpart F ("Promoting Objectivity in Research") for PHS/NIH grants and cooperative agreements, and 45 CFR Part 94 for research contracts. Note: These regulations do not apply to SBIR or STTR Phase I applications or awards.
Alchemie ("the Institution") adopts this policy for all Investigators engaged in PHS/NIH-funded research. It establishes processes to identify, disclose, and manage Investigator financial conflicts of interest to protect research integrity, ensure the safety of research subjects, and maintain public trust in federally supported research.
2. Applicability
This policy applies to all individuals who meet the regulatory definition of "Investigator" (defined in Section 3) who are planning to participate in, or who are participating in, PHS/NIH-funded research at Alchemie. This includes the Principal Investigator (PI), co-investigators, collaborators, consultants, and any other individuals responsible for the design, conduct, or reporting of PHS/NIH-funded research, regardless of title or position.
3. Definitions
Financial Conflict of Interest (FCOI): A Significant Financial Interest (SFI) that is related to PHS/NIH-funded research and could directly and significantly affect the design, conduct, or reporting of that research.
Financial Interest: Anything of monetary value, whether or not its value is readily ascertainable.
Significant Financial Interest (SFI): Defined in detail in Section 4. Generally, a financial interest of the Investigator (or their spouse or dependent children) that reasonably appears related to the Investigator's institutional responsibilities and meets applicable dollar thresholds.
Investigator: The PD/PI and any other person, regardless of title or position, responsible for the design, conduct, or reporting of PHS/NIH-funded research. Alchemie determines who qualifies based on role and degree of independence, not title alone.
Institutional Responsibilities: The professional activities an Investigator performs on behalf of Alchemie, including research, product and services development and testing, publication and communication of research, consulting, operations management, administration, fundraising, and committee memberships.
Designated Official (DO): The individual appointed by Alchemie to solicit and review SFI disclosures, determine whether FCOIs exist, and develop and oversee management plans. At Alchemie, this is the CEO or a designated senior staff member appointed by the CEO.
Manage: Taking action to address an FCOI, which can include reducing or eliminating the financial conflict of interest, to ensure research is conducted free from bias to the extent possible.
Senior/Key Personnel: The PD/PI and any other individual identified as senior/key personnel by Alchemie in a grant application, progress report, or other NIH submission.
PHS: The Public Health Service of the U.S. Department of Health and Human Services, including NIH.
Research: A systematic investigation designed to develop or contribute to generalizable knowledge, including basic and applied research and product development, supported by a PHS Awarding Component through a grant, cooperative agreement, or contract.
4. Significant Financial Interest (SFI)
4.1 What Constitutes an SFI
An SFI exists if the Investigator (or their spouse or dependent children) has any of the following interests that reasonably appear related to the Investigator's institutional responsibilities at Alchemie:
- Publicly traded entity: Total remuneration received in the previous 12 months plus the value of any equity interest exceeds $5,000.
- Non-publicly traded entity: Remuneration received in the previous 12 months exceeds $5,000, or the Investigator holds any equity interest.
- Intellectual property: Income from IP rights (patents, copyrights, royalties) exceeds $5,000 in the previous 12 months.
- Reimbursed or sponsored travel: Travel sponsored or reimbursed by an entity other than Alchemie, a U.S. federal/state/local government agency, a U.S. institution of higher education, an academic teaching hospital, a medical center, or a U.S.-affiliated research institute, with a value exceeding $5,000 in the previous 12 months. Disclosure must include purpose, sponsor, destination, and duration.
- Foreign financial interests: All financial interests from foreign entities must be disclosed regardless of amount, including remuneration, travel, advisory service income, and equity from foreign universities, companies, or governments.
4.2 What Does NOT Constitute an SFI
Investigators are not required to disclose the following:
- Salary, royalties, or remuneration paid by Alchemie to the Investigator for work performed at Alchemie, including IP rights assigned to Alchemie.
- Any ownership interest in Alchemie held by the Investigator (as Alchemie is a for-profit commercial entity).
- Income from investment vehicles such as mutual funds or retirement accounts where the Investigator does not directly control investment decisions.
- Income from seminars, lectures, or teaching engagements sponsored by a U.S. federal, state, or local government agency, a U.S. institution of higher education, an academic teaching hospital, a medical center, or a U.S.-affiliated research institute.
- Income from service on advisory committees or review panels for the same categories of institutions listed above.
5. Investigator Disclosure Requirements
5.1 When to Disclose
All Investigators must disclose their SFIs related to their institutional responsibilities at Alchemie — not limited to the specific funded research. Disclosures are required at the following times:
- At time of application: The PI and all Investigators must disclose SFIs to the DO before submission of a PHS/NIH proposal. Any new Investigator joining after submission must disclose promptly before participating.
- Annual disclosure: Each Investigator must submit an updated SFI disclosure on or before January 1 each year for the duration of the award, including new or updated information on previously disclosed SFIs.
- Within 30 days of new SFI: Investigators must disclose any new SFI (including through purchase, marriage, or inheritance) within 30 days of discovering or acquiring it. Updated disclosure of reimbursed or sponsored travel must also be submitted within 30 days of each occurrence.
5.2 How to Disclose
Investigators submit disclosures using the SFI Disclosure Form included at the end of this policy. Completed forms are submitted to the Designated Official. SFI disclosures are retained by Alchemie for at least three years from the date of submission of the final expenditures report.
6. Review of SFI Disclosures
The Designated Official (DO) reviews all SFI disclosures. If the DO is not available, the CEO or a designated consultant may fulfill this function.
The DO evaluates each disclosed SFI against every PHS/NIH research application or award on which the Investigator is responsible to determine: (1) whether the SFI is related to the funded research, and (2) if related, whether it constitutes an FCOI.
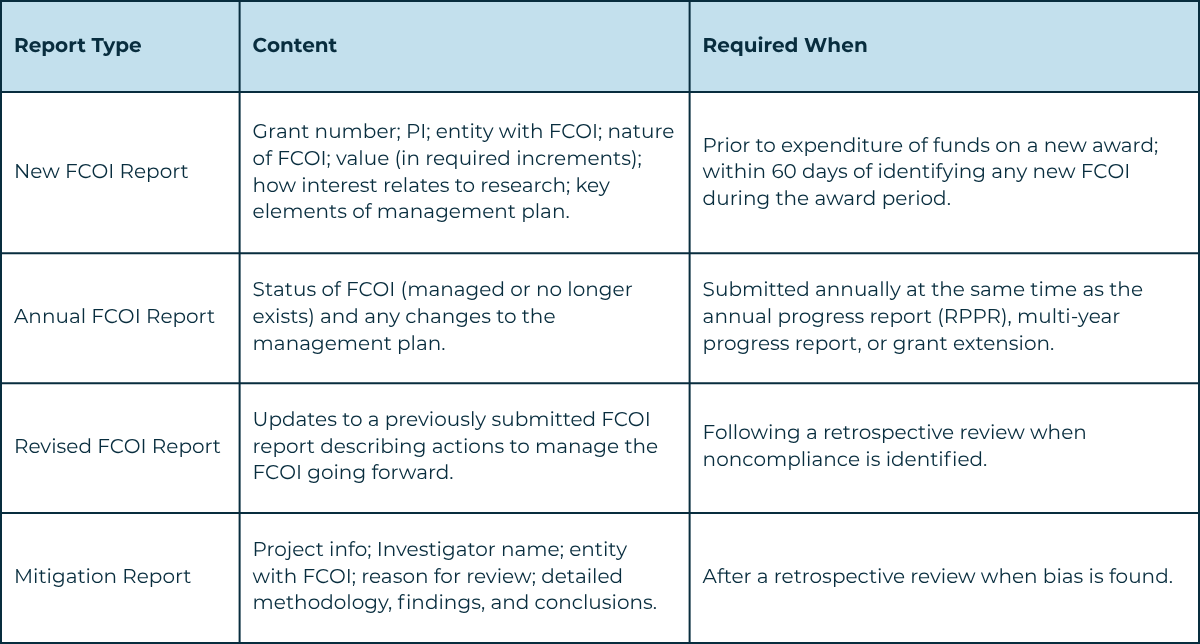
- Prior to award expenditure: The DO reviews all SFIs before NIH issues a new award. If an FCOI is identified, an FCOI report is submitted to NIH via the eRA Commons FCOI Module prior to any expenditure of funds.
- Annual review: As part of annual disclosures, the DO reviews updates for changes to existing SFIs and determines whether management plan modifications are needed.
- Ad hoc during award: If a new Investigator joins or an Investigator acquires a new SFI, the DO will within 60 days: review the disclosure; determine relatedness; determine if an FCOI exists; and if so, implement an interim management plan. An FCOI report will be submitted to NIH within 60 days.
7. FCOI Determination
An SFI is considered "related" to NIH-funded research when the DO reasonably determines that the SFI could be affected by the research, or that the SFI is in an entity whose financial interests could be affected by the research.
An FCOI exists when the DO determines that the related SFI could directly and significantly affect the design, conduct, or reporting of the PHS/NIH-funded research. The DO may consult with the Investigator during this assessment.
8. Management of FCOIs
When an FCOI is identified, the DO implements a management plan to ensure research is conducted objectively. Management strategies may include:
- Public disclosure of the FCOI in publications, presentations, or to study personnel, IRB, IACUC, or Data Safety Monitoring Board.
- For human subjects research, disclosure of the FCOI to participants in the informed consent document.
- Appointment of an independent monitor to protect against bias in the design, conduct, and reporting of the research.
- Modification of the research plan.
- Change of personnel roles or removal from portions of the research.
- Reduction or elimination of the financial interest (e.g., divesting equity).
- Severance of relationships creating the conflict.
The DO communicates the FCOI determination and management plan in writing to the Investigator, PD/PI, and appropriate supervisor. No funds may be expended on an NIH award until the Investigator has met all disclosure requirements and agreed in writing to comply with the management plan. The DO submits an FCOI report to NIH via the eRA Commons FCOI Module.
9. Monitoring Compliance
Alchemie monitors Investigator compliance with management plans for the duration of each NIH award. FCOIs must be disclosed in publications, presentations, and other communications. Investigators must also disclose the FCOI in writing to study personnel and provide a copy of this disclosure to the DO for recordkeeping.
10. NIH Reporting Requirements
10.1 FCOI Reports
Prior to spending any funds under a NIH-funded award, Alchemie submits an FCOI report to NIH for any Investigator SFI determined to be an FCOI. Alchemie designates an institutional official as the FCOI Signing Official (FCOI SO) in the eRA Commons FCOI Module.
11. Public Accessibility
A copy of this FCOI policy is publicly available on Alchemie's website at: alchem.ie/blog/fcoi, as required by NIH Grants Policy Statement Section 4.1.10.
Before any funds are spent under an NIH award, Alchemie will ensure public accessibility of identified FCOIs held by Senior/Key Personnel — either by website posting or written response within five (5) business days to requests. Public disclosures include: Investigator name and title, entity in which the SFI is held, nature of the SFI, and approximate dollar value in the required ranges.
This information is updated at least annually and within 60 days of any new FCOI identification. Records are maintained for at least three years from the most recent update.
12. Training Requirements
Each Investigator must complete FCOI training before engaging in PHS/NIH-funded research, at least once every four years, and promptly when any of the following occur:
- Alchemie revises this FCOI policy in a way that affects Investigator requirements.
- An Investigator is new to Alchemie research under an NIH award (training must be completed before participating).
- The DO determines that an Investigator has not complied with this policy or a management plan (training must be completed within 30 days as directed by the DO).
Investigators fulfill the training requirement by completing the NIH Financial Conflict of Interest tutorial at: https://grants.nih.gov/grants/policy/coi/tutorial2018/story_html5.html and reviewing the NIH Virtual Seminar on FCOI compliance at: https://www.youtube.com/watch?v=D292YZ6BX24. Training certificates must be shared with the DO and retained on file.
13. Noncompliance and Corrective Action
If Alchemie identifies an SFI that was not disclosed, reviewed, or managed in a timely manner, the DO will within 60 days: review the SFI; determine relatedness; determine if it constitutes an FCOI; and if so, implement an interim management plan and submit an FCOI report to NIH.
In cases of noncompliance, Alchemie will within 120 days conduct a retrospective review of the Investigator's activities to determine whether research was biased. If bias is found, Alchemie will promptly notify NIH and submit a mitigation report as required by 42 CFR 50.605(a)(3)(iii).
Compliance with this policy is a condition of employment and participation. Investigators who fail to comply may be subject to disciplinary action, including termination of employment or contract, restrictions on research fund use, and/or disqualification from PHS/NIH-funded research.
14. Clinical Research Requirements
If HHS determines that a PHS-funded clinical research project evaluating the safety or effectiveness of a drug, medical device, or treatment was designed, conducted, or reported by an Investigator with an unmanaged or unreported FCOI, Alchemie will require the Investigator to disclose the conflict in every public presentation of the research results and to request an addendum to previously published presentations.
15. Subrecipient Requirements
When Alchemie enters into a written agreement with a subrecipient (collaborator, consultant, subcontractor, or sub-awardee) on a PHS-funded project, Alchemie will specify in the agreement whether Alchemie's FCOI policy or the subrecipient's own compliant FCOI policy applies.
- If the subrecipient's policy applies: The subrecipient certifies compliance with federal FCOI regulations and agrees to report identified FCOIs to Alchemie within 50–55 days of identification, enabling Alchemie to meet NIH reporting deadlines.
- If the subrecipient cannot certify compliance: Alchemie's FCOI policy applies. Subrecipient Investigators disclose SFIs directly to Alchemie's DO, and Alchemie manages, monitors, and reports any FCOIs to NIH.
16. Records Maintenance
Alchemie retains records of all Investigator SFI disclosures, DO reviews, FCOI determinations, management plans, and all related actions for at least three years from the date of submission of the final expenditures report, or longer as required by 45 CFR 75.361.
17. Point of Contact
For questions about this policy, to submit a disclosure, or for FCOI-related inquiries, contact:
Julia Winter
CEO, Alchemie
julia@alchem.ie
18. NIH Resources
NIH FCOI inquiries: FCOICompliance@mail.nih.gov
FCOI Regulation: 42 CFR Part 50 Subpart F — https://www.ecfr.gov/current/title-42/part-50/subpart-F
NIH FCOI Policy: https://grants.nih.gov/grants/policy/coi/index.htm
FCOI Training Tutorial: https://grants.nih.gov/grants/policy/coi/tutorial2018/story_html5.html
NIH FAQs: https://grants.nih.gov/faqs#/financial-conflict-of-interest.html
eRA Commons FCOI Module User Guide: https://www.era.nih.gov/files/fcoi_user_guide.pdf
19. Revision History
Version: 1.0
Date: 4-3-2026
Author: Julia Winter
Description: Initial version – for website
